Tag Archives: chemistry
The Chemistry of Champagne – Reactions
Do Carrots Help You See Better?
The Chemistry of Pizza – Reactions
A Cup Of Chemistry

Here’s for All the Coffee Lovers Out There
Thank You All

“As we express our gratitude, we must never forget that the highest appreciation is not to utter words, but to live by them.” – JFK
Special Thanks to my Family and Friends for your Kind Words of Encouragment and Support…I thank God for the Strength, Knowledge and Wisdom to take on such a Task of Informing the Society on the Fascinating World of Biochemistry… I am much indebted to my lecturer for all the critism and knowledge of the subject matter…
Lastly I Thank You All My Bloggers/Followers for Your Continued Support…Words Cannot Express My Gratitude…
Wordle #2 Glycolysis

Words involved in the process of Glycolysis!
Does One Really Know What Is Arsenic Poisoning??? Lets Learn More…
While doing some research I came across this very interesting article entitled “Arsenic Poisoning”, discussant Michael S. Gorby, M.D, Albuquerque. This article may seem a bit miss leading at first; I actually thought this article was based on two patients who had experienced arsenic poisoning. A 17 years old woman whom attempted suicide by ingesting arsenic poisoning trioxide containing rodenticide and 64 year old man who suffered with chronic arsenic poisoning who was admitted several times in hospitals for various complications.
However, this article instead discussed the chemistry of the element Arsenic, the sources of arsenic poisoning: it’s natural, industrial and administered sources. Also the biochemistry and toxicity of arsenic, the effects of arsenic poisoning whether it is acute or chronic and the treatment of arsenic poisoning were discussed.
So What is Arsenic Poisoning???
 http://sp.life123.com/bm.pix/arsenic.s600x600.jpg
http://sp.life123.com/bm.pix/arsenic.s600x600.jpg
Arsenic is a member of the nitrogen family or group 5 of the periodic table of the elements. It has an atomic number 33 with an atomic weight of 74.9 and is classified as a transition element or metalloid. Arsenic is a natural element found in soil and minerals. Arsenic compounds are used to preserve wood, as pesticides, and in some industries. Arsenic can get into air, water, and the ground from wind-blown dust. It may also get into water from runoff.
You may be exposed to arsenic by:
- Taking in small amounts in food, water or air
- Breathing sawdust or burning smoke from arsenic-treated wood
- Living in an area with high levels of arsenic in rock
- Working in a job where arsenic is made or used
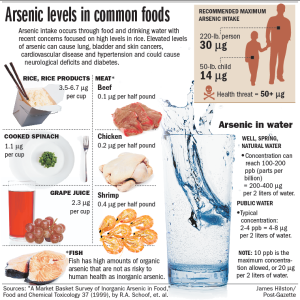 http://e-infopages.com/wp-content/uploads/2012/11/arsenic_in_food.png
http://e-infopages.com/wp-content/uploads/2012/11/arsenic_in_food.png
Exposure to arsenic can cause many health problems. Being exposed to low levels for a long time can change the color of your skin. It can cause corns and small warts. Exposure to high levels of arsenic can cause death.
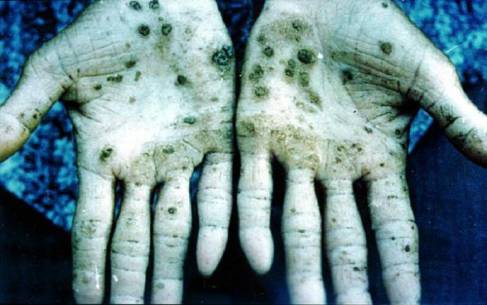 http://www.ualberta.ca/~xcle/img/hands.jpg\
http://www.ualberta.ca/~xcle/img/hands.jpg\
REFERENCE
Gorby.S. Michael. 1988. Arsenic Poisoning. US National Library of Medicine. National Institute of Health. Western Journal of Medicine. Acessed 20th March, 2013. 308-315. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1026413/
What type of non-covalent interaction helps to stabilize the secondary structure of proteins?
ANSWER
Hydrogen Bonding
Regardless of whether or not the secondary structure is a beta sheet or alpha helix, the two main portions of the peptide bond (the carbonyl portion and the amino portion) on different amino acids orient themselves so that so that the hydrogen of the amino portion on one amino acid hydrogen bonds to the oxygen of the carbonyl portion of another. This essentially stabilizes the structure.






